Abstract
Background:
Venetoclax-based combinations were recently approved to treat patients (pts) with acute myeloid leukemia (AML) ineligible for intensive chemotherapy. Limited prospective 'real-world' data is available on treatment patterns of venetoclax-based therapy in routine clinical practice. We investigated patterns of patient selection, efficacy, toxicity, patient related outcome and post-remission management in a nationwide multicenter prospective observational trial.
Methods:
Newly diagnosed pts with AML were enrolled at the time of venetoclax-based therapy initiation from 10 medical centers in Israel. Demographic, clinical and patient-related baseline characteristics were documented. Treatment patterns, safety and efficacy outcomes are reported.
Results:
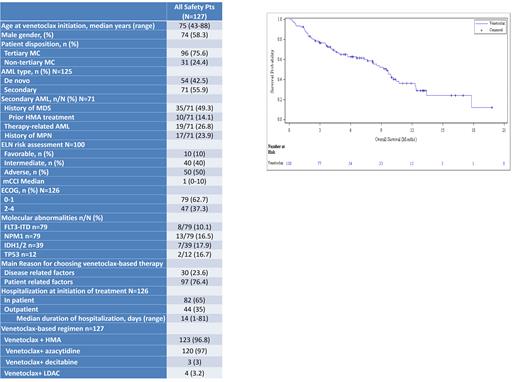
Between August 12, 2019, and June 17, 2021(data cut) ,127 AML pts were enrolled to receive venetoclax based therapy. Baseline patient and disease characteristics are reported in Table 1.
The main reasons for physician's choice of venetoclax-based therapy were age ≥75, comorbidities and ECOG ≥2 (patient related factors) in 76% of cases and adverse disease biology predicting poor response to intensive chemotherapy (disease related factors) in 24% of cases. Most pts started therapy in an inpatient setting, 82 (64.6%) with a median hospitalization duration of 14 days, while 44 pts (34.6%) started therapy as out pts.
Pts received a median of 3.8 cycles of therapy (range 1-21). Most pts (97%) received venetoclax in combination with hypomethylating agents. The full dose of 400mg QD after a median ramp-up duration of 3 days was achieved in 88% of the pts. Dose interruptions and dose modifications during follow-up occurred in 59 (46%) and 30 (24%) of pts, respectively.
To allow for adequate follow up for response assessment, efficacy analysis was limited to pts enrolled prior to December 31, 2020, and included 108 pts with a median follow-up of 8 months (range 1-20). As of data cut, 93 pts completed cycle 1 of therapy, 66 pts completed cycle 3 and 39 pts completed cycle 6. 29 pts (27%) are still active on treatment. Best composite complete remission [CCR = complete remission (CR) plus CR with incomplete count recovery (CRi)] was achieved in 62 (57%) pts. CCR rates were assessed in different pre-defined subgroups. Best CCR in pts selected for therapy based on disease-related and patient-related factors were 70% and 54% respectively. Best CCR in pts with AML arising from MPN and pts with other AML were 45% and 58% respectively.
Estimated median overall survival (OS) of all pts was 9.6 months (range 7.4-10.6) (Figure 1). Achieving CCR was associated with a superior probability for survival. Estimated median OS was 13.6 months (range 10.6 - not reached) in pts achieving CCR and 4.2 months (range 1.2-10.3) in non-CCR (p<.0001). Of responding pts (CR/CRi, partial remission (PR), morphologic leukemia free state (MLFS), 27 (37%) progressed. Estimated median time to progression was 9.2 months (6.7-NR).
Allogeneic transplantation following venetoclax based treatment was offered to 16 (26%) pts with a median age of 71 years (range 43-77). Last documented response prior to transplant was CR in 5 (32%) pts, CRi 9 (56%), MLFS 1 (6%) and PR in 1 (6%) patient.
Among grade ≥3 AEs were febrile neutropenia in 28% and infections in 21% of pts. Clinical and laboratory tumor lysis syndrome (TLS) was documented in 2 and 4 pts, respectively. Antifungal prophylaxis was administered in 20% of pts and granulocyte colony-stimulating factor (GCSF) support was used in 17% of pts in response. Early death rate at 30 and 60 days were 7% and 13%, respectively.
Conclusion:
This prospective real-world analysis reveals unique patterns of patient selection and venetoclax treatment utilization in a medical system with wide access for this indication. Venetoclax-based therapies are effective and associated with manageable toxicity, including in AML patient populations that were excluded from previous registration trials with comparable CCR and early death rates. Factors associated with patient selection in the 'real-world' setting and immature follow up data most probably led to a shorter estimated median OS in this analysis as compared to controlled trials.
The REVIVE study continues to expand and is expected to provide additional insights on treatment patterns, management as well as clinical and patient related outcomes.
Wolach: Janssen: Consultancy; Novartis: Consultancy; Amgen: Research Funding; Astellas: Consultancy; Abbvie: Consultancy, Honoraria, Research Funding; Neopharm: Consultancy. Levi: AbbVie: Consultancy, Research Funding. Lavie: AbbVie: Membership on an entity's Board of Directors or advisory committees, Other: Fees for lectures; BMS: Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Other: Fees for lectures; Roche: Other: Fees for lectures; Novartis: Other: Fees for lectures. Tavor: AbbVie: Consultancy. Hellmann: AbbVie: Consultancy. Tadmor: Janssen: Consultancy, Honoraria, Research Funding; AbbVie: Consultancy, Honoraria, Research Funding. Zuckerman: Gilead Sciences: Honoraria, Speakers Bureau; Novartis: Honoraria; Janssen: Honoraria; Cellect Biotechnology: Honoraria; BioSight Ltd: Honoraria; AbbVie: Honoraria; Orgenesis Inc.: Honoraria. Stemer: AbbVie: Consultancy. Berelovich: AbbVie: Current Employment, Current equity holder in publicly-traded company. Ofek: AbbVie: Current Employment, Current equity holder in publicly-traded company. Frankel: AbbVie: Current Employment, Current equity holder in publicly-traded company. Grunspan: AbbVie: Current Employment, Other: May hold equity. Ofran: Medison Israel: Consultancy; Pfizer: Consultancy; Astellas: Consultancy; AbbVie: Consultancy; Janssen: Consultancy. Moshe: Novartis: Membership on an entity's Board of Directors or advisory committees, Other: Lectures; Astellas: Membership on an entity's Board of Directors or advisory committees, Other: Lectures; AbbVie: Membership on an entity's Board of Directors or advisory committees, Other: Lectures.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal